Peer Reviewed
Langerhans Cell Histiocytosis Presenting as Pneumothorax and Bleeding Gums in a Pediatric Patient
Author:
Anupam Mittal, DO
Baylor Scott & White Medical Center, Round Rock, Texas
Citation:
Mittal A. Langerhans cell histiocytosis presenting as pneumothorax and bleeding gums in a pediatric patient. Consultant. Published online August 12, 2019.
A 3-year-old boy with no medical history presented to the emergency department (ED) for worsening cough, decreased appetite, tachypnea, and chest pain for 2 nights.
The mother’s prenatal course and delivery had been normal. Neonatal hearing, metabolic, and congenital heart defect screenings were negative. There had been no preceding trauma or significant illnesses. The patient had visited a dentist for “bleeding gums” 2 weeks prior.
At presentation in the ED, the patient’s vital signs were normal. Physical examination findings were unremarkable except for absent right-sided breath sounds and abdominal retractions. Chest radiographs showed a spontaneous right tension pneumothorax (Figure 1). A chest tube was placed, and a computed tomography (CT) scan of the chest was ordered (Figure 2), the latter of which showed bilateral diffuse cystic lesions leading to honeycombing. The patient was admitted for further workup.
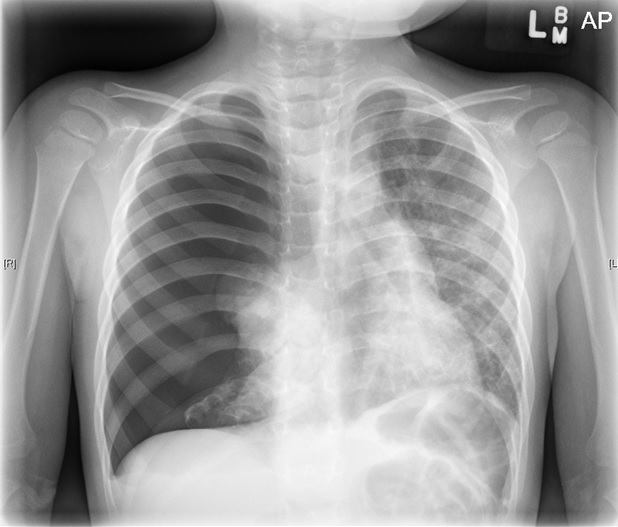
Figure 1. Chest radiograph showed a spontaneous right tension pneumothorax.

Figure 2. CT scan showed diffuse bilateral cystic disease after chest tube placement.
The differential diagnosis included Langerhans cell histiocytosis (LCH), cystic fibrosis (CF), and congenital etiologies. A pulmonologist consultant felt that CF was unlikely given the lack of failure to thrive, the lack of prior findings, and the normal neonatal metabolic screening results. LCH was considered less likely given the isolated pulmonary involvement without known bone or skin lesions. A hematologist-oncologist was consulted, and video-assisted thoracoscopic surgery (VATS) lung biopsy with bronchial-alveolar lavage was planned. Results of an osseous radiological survey were negative for abnormalities.
The panoramic dental radiograph from the patient’s dentist visit 2 weeks earlier was obtained (Figure 3), and an oral mucosal biopsy was performed after concerns about possible abnormal soft tissue in the gingiva and lytic lesions around the mandibular molars. CT scan of the mandible, performed in the hospital after these images were received, confirmed mandibular lytic lesions.
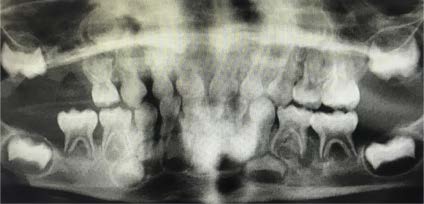
Figure 3. Panoramic dental radiograph taken at a dentist visit 2 weeks before presentation showed poorly visualized mandibular lytic lesions.
The biopsy revealed the presence of LCH, so VATS biopsy and bronchoscopy were not performed. Echocardiography was ordered to evaluate for pulmonary hypertension, and diagnostic bone marrow studies were ordered.
CT scans of the mandible and noncontrast panoramic dental radiographs showed periodontal osseous erosions and lytic lesions of the mandibular molars and osseous erosion at the dental fossa base, possibly representing an early oral antral fistula. Magnetic resonance imaging of the brain was performed, the results of which were negative for lesions suspicious of LCH but showed mild paranasal sinus disease and opacification of the mastoid air cells. Echocardiography findings were normal.
The patient underwent chest tube replacement, port placement, bone marrow biopsy, and oral surgery for extraction of nonviable teeth. Chemotherapy with vinblastine and prednisone was started.
Discussion. Spontaneous pneumothorax (SP) is rare and may have nonspecific presentations in children. SP is defined as a rupture of visceral pleura with air accumulation within the pleural space, which can be primary or secondary. Tension pneumothorax (TP) creates increased intrathoracic pressure, leading to mediastinal shift, respiratory distress, hypoxia, and hemodynamic instability, as illustrated in Figure 1.1
Among the causes of secondary SP are CF, asthma, and interstitial lung disease.1 The prevalence of pediatric primary spontaneous pneumothorax (PSP) is less than 0.01% in the general population,2 and the condition predominates in males, smokers, newborns, and adults.3
SP usually presents at rest or with minimal exertion with sudden onset of symptoms. Chest radiographs should be obtained in the upright position; inspiratory films are preferable. Chest CT scanning is obtained for chest tube placement and to help determine etiology.4,5
Pediatric SP has many differential diagnoses including simple viral, bacterial, or fungal infections; CF; LCH; Marfan syndrome; and malignancy.7,10,11 LCH can present in many ways, including SP. Lung findings can be seen in the absence of clinical pulmonary symptoms, so it is vital to be familiar with alternative presentations, such as in bone as illustrated in Figure 3.9,10 If there is enough suspicion to warrant imaging, a CT scan can provide clues to anticipate future episodes of PSP.8 Counseling on long-term management of any lung pathology in children is warranted.6 Smoking cessation, as evidenced among pregnant women with LCH, appears to be a critical factor impacting the course of the disease.6 Follow-up and long-term prognosis of SP is dependent on etiology, and treatments will differ accordingly.
References:
- Sahn SA, Heffner JE. Spontaneous pneumothorax. N Engl J Med. 2000; 342(12):868-874.
- Dotson K, Johnson LH. Pediatric spontaneous pneumothorax. Pediatric Emerg Care. 2012;28(7):715-720.
- Bense L. Spontaneous pneumothorax. Chest. 1992;101(4):891-892.
- O’Connor AR, Morgan WE. Radiologic review of pneumothorax. BMJ. 2005;330(7506):1493-1497.
- Choudhary AK, Sellars MEK, Wallis C, Cohen G, McHugh K. Primary spontaneous pneumothorax in children: the role of CT in guiding management. Clin Radiol. 2005;60(4):508-511.
- Radzikowska E, Wiatr E, Franczuk M, Bestry I, Roszkowski-Śliż K. Lung function in pregnancy in Langerhans cell histiocytosis. Adv Exp Med Biol. 2018;1023:73-83.
- Asilsoy S, Yazici N, Demir S, Erbay A, Koçer E, Sarıalioğlu F. A different cause for respiratory disorder in children: cases with pulmonary Langerhans cell histiocytosis. Clin Respir J. 2017;11(2):193-199.
- Kobayashi NS, Nambu A, Kawamoto M, et al. Pulmonary apical opacities on thin-section computed tomography: relationship to primary spontaneous pneumothorax in young male patients and corresponding histopathologic findings. J Comput Assist Tomogr. 2018;42(1):33-3
- Tang X, Wang W, Liu J, Yang H, Zhao S, Li H. Clinical characteristics of children with Langerhans cell histiocytosis with pulmonary involvement [in Chinese]. Zhonghua Er Ke Za Zhi. 2014;52(12):902-905.
- Martínez García JJ, Rios Osuna MG, Altamirano Álvarez E. Bilateral spontaneous pneumothorax as a setting of Langerhans cell histiocytosis. Arch Argent Pediatr. 2014;112(3):e113-e116.
- Varkki S, Tergestina M, Bhonsle VS, Moses PD, Mathai J, Korula S. Isolated pulmonary Langerhans cell histiocytosis. Indian J Pediatr. 2013;80(8):700-703


